
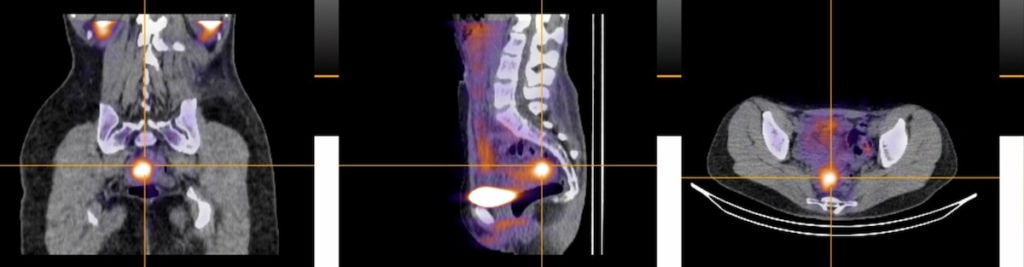
Serac Healthcare and the University of Oxford have published Phase 2 data in The Lancet Obstetrics, Gynaecology & Women’s Health showing that 99mTc-maraciclatide, a molecular imaging agent, can visualize endometriosis non-invasively – including superficial peritoneal endometriosis (SPE), the most common subtype that currently requires laparoscopic surgery for definitive diagnosis because conventional imaging can’t reliably detect it.
In the DETECT study, imaging results were concordant with the surgical presence or absence of endometriosis in 16 of 19 cases, with endometriosis detected in 14 of 17 surgically positive participants. No false positives were reported. Notably, 10 participants with SPE had undergone conventional imaging (transvaginal ultrasound and/or MRI) within the previous 12 months, and none of those scans had detected SPE.
The imaging agent works by binding to αvβ3 integrin, which is upregulated during angiogenesis – the formation of new blood vessels that is a hallmark of inflammatory diseases including endometriosis. Beyond diagnosis, the researchers highlight its potential as a tool for monitoring disease recurrence and measuring treatment response, which could support the development of new endometriosis therapies.
“Novel, non-invasive diagnostic tests for endometriosis are a global research priority,” said Professor Christian Becker, co-director of the Endometriosis CaRe Centre in Oxford. “If these Phase II results are reproduced in the Phase 3 studies, maraciclatide has the potential to be an extremely valuable tool, as it could both reduce diagnostic delays and provide a validated endpoint for the development of new therapeutics.”
“Superficial peritoneal endometriosis, the most prevalent subtype of endometriosis, currently evades reliable detection, leaving women no choice for diagnosis other than invasive surgery,” said Professor Krina Zondervan, head of department at Oxford’s Nuffield Department of Women’s and Reproductive Health. “If these results are confirmed in larger Phase 3 studies, imaging with maraciclatide could transform clinical research and practice and potentially empower the development of treatments for women across the globe.”
SPE accounts for 80% of all laparoscopically diagnosed endometriosis, making a non-invasive detection method for this subtype particularly significant. Serac Healthcare has FDA Fast Track Designation and agreed Phase 3 study designs, with multi-center international Phase 3 trials planned to begin later this year. The DETECT study was funded by the NIHR Oxford Biomedical Research Centre and Serac Healthcare.


