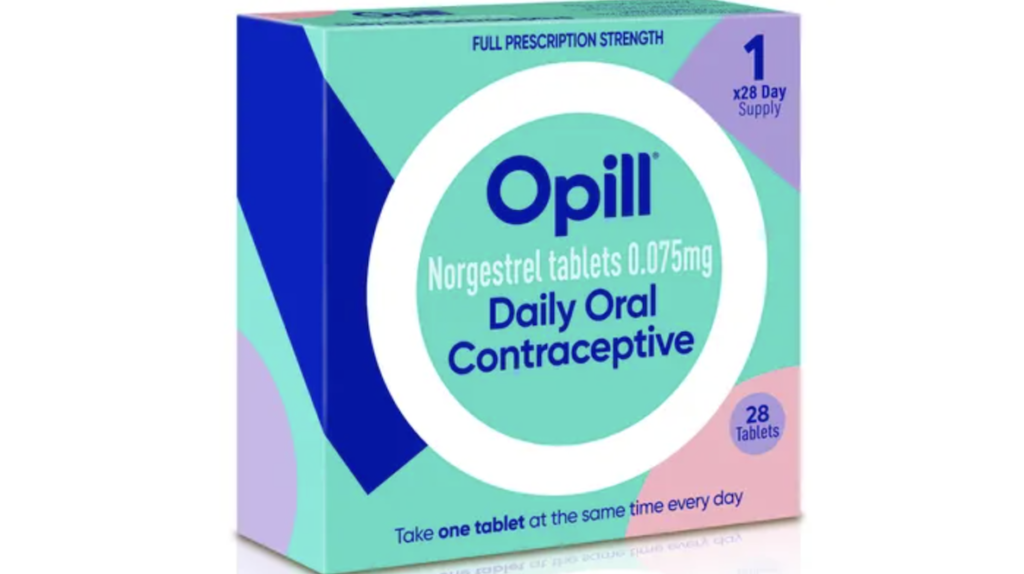
Perrigo has achieved a major milestone in women’s healthcare with the FDA approving Opill as an over-the-counter oral contraceptive. Opill is the first birth control pill available without a prescription in the US, revolutionizing access to contraception. Perrigo’s commitment to enhancing lives is evident in this impactful solution for women’s health.
Opill has a long history of safe and effective use, supported by scientific evidence and endorsed by major medical organizations. This achievement marks a significant leap for women’s empowerment, improving access to contraception and addressing a crucial need.
“Today’s approval is a groundbreaking expansion for women’s health in the U.S., and a significant milestone towards addressing a key unmet need for contraceptive access,” said Frederique Welgryn, Perrigo Global Vice President for Women’s Health. “Perrigo is committed to making Opill, which is now the most effective method available OTC at preventing pregnancy, accessible and affordable to women and people of all ages. I want to thank the FDA and its advisory panels, the Free The Pill coalition, the numerous medical organizations and advocacy groups, and all those who wholeheartedly supported this enormous undertaking that has made today a giant leap for women’s empowerment.”
The unanimous support of the FDA’s advisory committees underscores the benefits of OTC access outweighing any associated risks. Opill will be available both in-store and online in early 2024.



