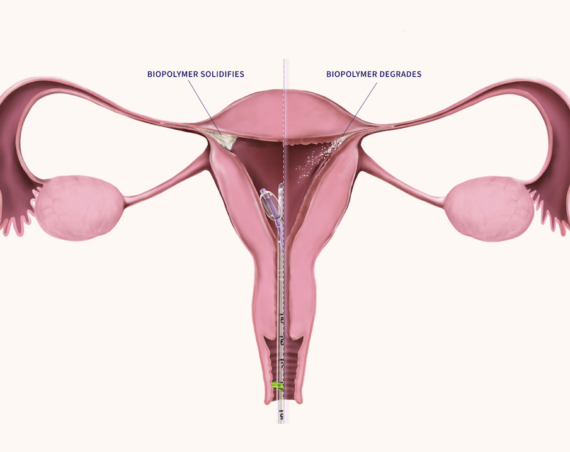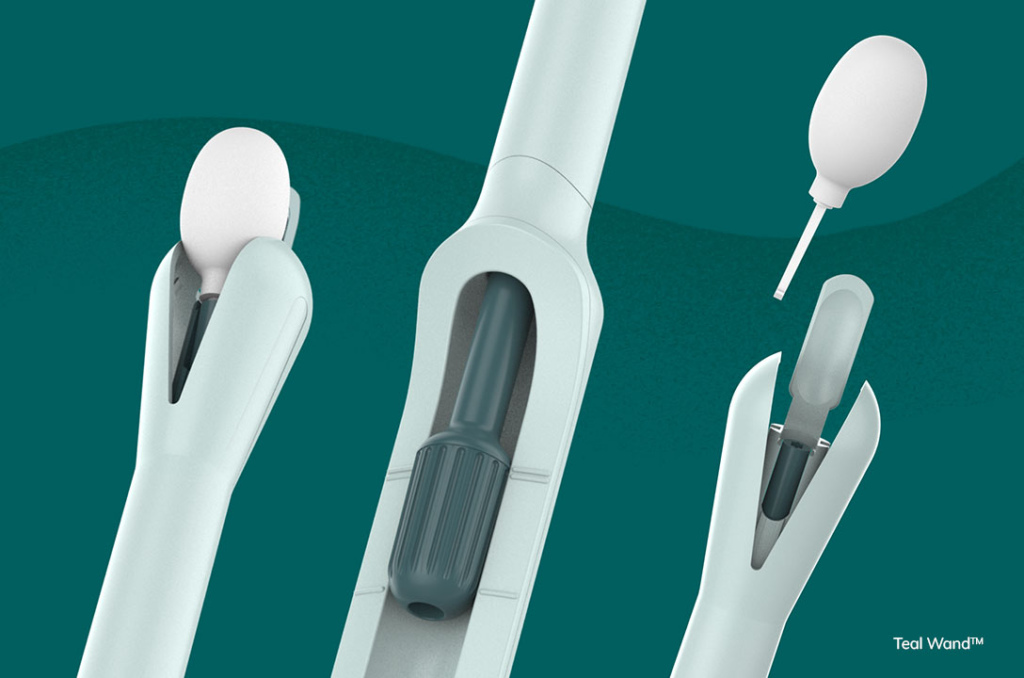
Teal Health is making strides in cervical cancer prevention with the SELF-CERV clinical study of its Teal Wand, an at-home self-collect device for cervical cancer screening. The study, underway at top health institutions such as Johns Hopkins, Yale University, and the University of Colorado, supports FDA submission and approval of the device. Teal Health’s initiative aims to provide a convenient and less invasive screening option for the 25% of US women who are not current with their cervical cancer screenings.
Cervical cancer, while nearly 100% preventable with early detection, continues to affect over 12,000 individuals annually in the US, with more than half of the diagnoses at a later stage. The Teal Wand offers a simple, comfortable solution for individuals to collect their own vaginal sample at home or in a health clinic, addressing barriers such as discomfort during exams, lack of gynecological care access, and limited health literacy.
The Teal Health clinical study includes participation from more than 15 health organizations, like multiple Planned Parenthood Research Network locations and Woman’s Hospital in Baton Rouge. Goal of the study is to evaluate the effectiveness of patient self-collection compared to clinician collection for detecting high-risk HPV, responsible for over 90% of cervical cancers. With HPV testing as the recommended cervical cancer screening method, the study’s outcomes could significantly impact screening adherence and overall women’s health.
“Through my medical practice as the director of the complex dysplasia clinic, working to increase screening rates is a top concern,” said Dr. Christine Conageski, Associate Professor and OB-GYN at the University of Colorado Anschutz Medical Campus. “With national screening rates plateauing or even declining in areas, it requires new approaches to make significant progress. At-home screening options will provide a great alternative to the in-person traditional speculum exam and will increase access and address screening barriers, specifically the need for a more convenient and comfortable experience for women.”
This clinical study follows an initial study that included over 220 participants ranging in age, race, socioeconomic status, and education across multiple states and regions. The study demonstrated that not only did participants successfully self-collect a sample, but that it was much preferred to the speculum – 92% said if they knew the results were equal they would prefer self-collection, and 87% reported they would stay current with screening if Teal Wand was an option. Self-collection took the vast majority under 2 minutes to complete, and 97% of study participants reported it was very easy or easy to use.
Elizabeth Sutton, PhD, Principal Investigator from the Woman’s Hospital in Baton Rouge, Louisiana says, “Women in our community experience many barriers when it comes to accessing care. What we’ve loved about working with Teal in the initial study and now moving into the clinical trial is the excitement by the participants to be a part of the study, and to have their input and experience matter.”
“Self-collection is now a viable option thanks to advancements with primary HPV assays, mirroring practices seen in other parts of the world,” said Kara Egan, CEO and Co-founder of Teal Health. “The self-collection approach enables us to revolutionize the screening process and design it for the modern woman. We’re thrilled to have such incredible partners and participants help us execute on this life-saving screening option in the United States, prioritizing and respecting a woman’s needs, time, and preferred experience.”